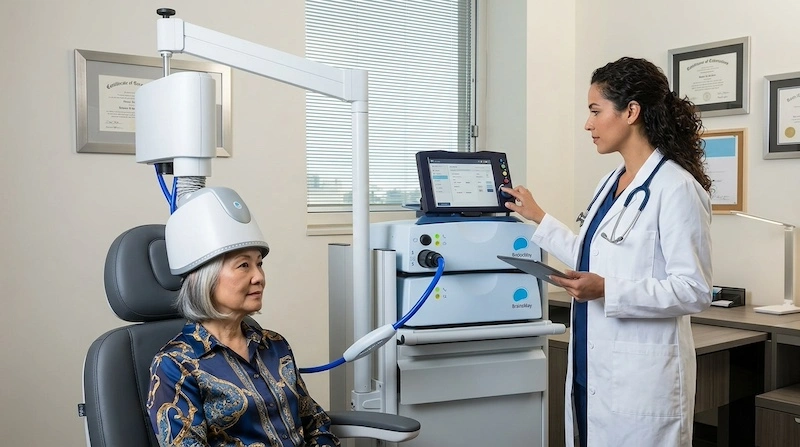
According to a 2024 open-label case series published in Brain Stimulation (ScienceDirect, Q4 2024), 32 patients with treatment-resistant depression who completed a single-day TMS regimen showed a 90% response rate at six weeks across multiple validated depression and anxiety scales.
That finding raises an obvious question for anyone who has been putting off TMS therapy: what if weeks of treatment could be compressed into a single day?
This article explains what the one day TMS treatment entails, how it compares to standard protocols, what the current research shows, and how to decide whether it belongs in your treatment conversation.
What Is One Day TMS?
One day TMS is an accelerated treatment protocol that delivers multiple TMS sessions in rapid sequence within a single clinic visit, rather than spreading them across four to six weeks. Think of it as the difference between commuting daily for a month versus completing the journey in one long trip. The approach emerged from mounting evidence that the brain can tolerate and respond to more stimulation per day than traditional schedules assumed.
How Does Standard TMS Differ From a One Day TMS Treatment?
Standard TMS, cleared by the FDA in 2008, involves one session per day, five days a week, for four to six weeks. Each session delivers roughly 3,000 magnetic pulses over 20 to 40 minutes, adding up to 60,000 to 90,000 total pulses across 20 to 30 visits. The schedule works well for many patients, but the sheer number of appointments creates real barriers: time off work, transportation costs, arranging childcare repeatedly, or simply living far from a clinic.
One day TMS collapses that schedule. Instead of 30 separate visits, everything is delivered in a single, extended appointment.
What the ONE-D Protocol Actually Involves
The most studied single-day framework is the ONE-D (Optimized, Neuroplasticity-Enhanced techniques in Depression) protocol. ONE-D condenses treatment for major depressive disorder and generalized anxiety disorder into a single day, delivering 20 high-frequency iTBS sessions (600 pulses each, at 120% motor threshold) over approximately 9.5 hours. Sessions are spaced roughly 30 minutes apart, which research suggests is the optimal interval for preserving the neuroplasticity effects between rounds.
The term One-D TMS protocol is sometimes used interchangeably with ONE-D TMS, and you may also see it written as one-day TMS treatment in patient-facing materials. They all refer to the same core idea: compressing the full therapeutic dose into one calendar day.
Some versions of the protocol also incorporate pharmacological agents designed to enhance the brain’s plasticity response. This is an active area of study, and not all clinics offering one day TMS include this component.
What Does the Research Say About the ONE-D TMS Protocol?
The evidence base for one day TMS protocol is newer than that for standard TMS, and it’s important to read it with the appropriate context. That said, early results are notable.
A retrospective case series examining the ONE-D regimen found that at 26 weeks, 50% of the 32 participants showed sustained remission on validated depression scales, while relapse or retreatment had occurred in approximately 25 to 28% of patients. For a group that had already failed multiple medication trials, those numbers represent meaningful outcomes.
For context on where TMS stands more broadly: a 2024 multi-center randomized controlled trial published in the American Journal of Psychiatry found that patients assigned to TMS showed response rates of 37.5% versus 14.6% for those switched to a different medication, and remission rates of 27.1% versus 4.9% respectively. One day TMS aims to achieve comparable results in a fraction of the time.
What Research Is Still Evolving
True single-day TMS protocols are newer, and the published evidence is not yet at the same level as longer accelerated approaches. Most one-day data comes from clinic-based observational reports rather than sham-controlled randomized trials. The core neuroscience is sound, but questions remain around optimal pulse totals, ideal inter-session intervals, and whether durability outcomes differ meaningfully from five-day accelerated approaches.
This is not a reason to dismiss one day TMS, but it is a reason to discuss it carefully with a qualified provider.
Who Might Be a Good Candidate for One Day TMS?
Not every patient is the right fit for this format, and the protocol itself may not be available at every TMS clinic. That said, a clear profile has emerged from the early literature.
Patients who tend to benefit most often share one or more of these characteristics:
- Prior medication failures: One day TMS, like all TMS, is particularly relevant for treatment-resistant depression where antidepressants have not produced adequate relief
- Scheduling barriers: Professionals with demanding work schedules, caregivers, or people with limited time off who cannot realistically commit to 30 separate appointments
- Geographic distance: Patients who live or work far from a TMS clinic and cannot make daily trips feasible
- Motivation for a defined endpoint: Some patients find the psychological clarity of a single-day commitment easier to approach than an open-ended six-week regimen
Patients with certain metal implants in or near the head, active seizure disorders, or other specific contraindications are generally excluded from TMS of any format. A thorough clinical evaluation always precedes any TMS treatment.
How Does One Day TMS Compare to Other Accelerated TMS Options?
Accelerated TMS covers a spectrum of approaches, and one day TMS sits at the compressed end of that range. The table below outlines how the main formats differ:
| Protocol | Sessions | Duration | FDA Status |
| Standard TMS (rTMS) | 20-30 sessions | 4-6 weeks | FDA cleared (2008) |
| iTBS (Theta Burst) | 20-30 sessions | 3-6 weeks | FDA cleared (2018) |
| SAINT (Stanford) | 50 sessions | 5 days | Cleared for TRD (2022) |
| One Day TMS (ONE-D) | 20 sessions | 1 day | Investigational |
The SAINT protocol, developed at Stanford, is often cited alongside one day TMS. SAINT involves 50 sessions administered over five days and requires a functional MRI for precise targeting, using 90,000 total stimulation pulses. ONE-D uses heuristic targeting similar to standard TMS, requires no imaging, and delivers 12,000 pulses. One day TMS trades some of SAINT’s intensity for dramatically lower logistical complexity.
What Are the Risks and Limitations to Consider?
One day TMS is not a guaranteed solution, and any honest discussion of it has to include this section.
The limitations worth knowing before you pursue it:
- Evidence level: The ONE-D protocol is currently off-label in the United States. While early data is encouraging, large-scale randomized controlled trials have not yet been completed. Patients considering it should understand they would be receiving a protocol that is still building its evidence base.
- Response is not universal: Even in the most favorable published data, a meaningful proportion of patients do not respond or experience relapse within six months. Relapse or retreatment occurred in approximately 25 to 28% of participants at the 26-week mark. Managing expectations honestly matters here.
- Insurance coverage: Standard TMS is covered by most major insurers for qualifying diagnoses. One day TMS, given its off-label status, is unlikely to be covered in the same way. Out-of-pocket cost is a real consideration.
- Availability: Because the protocol requires extended clinic hours and specific expertise, not all TMS providers offer it. Asking a clinic whether they offer the ONE-D TMS protocol specifically is the right starting question.
- Physical demands of a long day: Sitting through 9 to 10 hours of treatment, even with rest between sessions, is taxing. Patients with certain health conditions may find this format more difficult than a shorter daily session.
If you’re weighing options for treatment-resistant depression,learn more about how TMS therapy works for depression at LifeQuality TMS before making any decisions.
How to Have a Productive Conversation With Your Provider About One Day TMS
Bringing this topic to your next psychiatric appointment does not require you to arrive with all the answers. It requires the right questions.
- Ask about your diagnosis fit: One day TMS has been studied primarily in major depressive disorder and generalized anxiety disorder. Ask your provider whether your specific diagnosis and history align with the patients in the published case series.
- Ask about the clinic’s experience: There’s a difference between a clinic that has read about one day TMS and one that has actually administered the ONE-D protocol. Ask how many patients they have treated this way and what outcomes they have observed.
- Request a comparison: Ask your provider to walk through how one day TMS compares to standard TMS therapy for someone with your specific history. The answer will be more informative than any article.
- Clarify the cost: Get a clear picture of what the session will cost out of pocket and whether any portion might be submitted to insurance for partial reimbursement.
- Ask about follow-up: Understand what happens after the single day. Is there a booster session protocol if response is partial? What does monitoring look like over the following weeks?
The Single Most Important Takeaway
One day TMS represents a genuine evolution in how TMS can be delivered, not a shortcut around the science that makes TMS effective. The neuroplasticity mechanisms that drive standard TMS are the same ones being targeted in the ONE-D TMS protocol. What changes is the delivery schedule, not the fundamental biology.
For patients who have hesitated to start traditional TMS because six weeks of daily appointments felt impossible, one day TMS offers a path worth exploring. It comes with caveats about evidence maturity and cost that deserve honest weight. But for the right patient, the question is no longer whether TMS is effective; it’s whether this particular format of it fits your life.
If you’re exploring TMS as an option for depression, anxiety, or OCD in the Brooklyn or New York City area, contact LifeQuality TMS to schedule a consultation and discuss which treatment approach may be appropriate for your situation.
Frequently Asked Questions
What is one day TMS and how is it different from regular TMS?
One day TMS is an accelerated treatment protocol that delivers a full course of TMS therapy, typically 20 sessions, within a single clinic visit lasting approximately 9 to 10 hours. Regular TMS spreads those sessions across four to six weeks with one appointment per day. The stimulation technology is the same; the delivery schedule is what changes.
Is the ONE-D TMS protocol FDA approved?
As of 2025, the ONE-D TMS protocol is considered off-label in the United States. Standard TMS (rTMS) was cleared by the FDA in 2008, and iTBS in 2018, but neither approval specifically covers a single-day compressed format. Patients considering this approach should discuss its investigational status with their provider.
Will one day TMS work if antidepressants haven’t helped me?
Early research on the ONE-D TMS protocol has been conducted specifically in populations with treatment-resistant depression, meaning patients who have already tried and not adequately responded to medications. Results in this group have been promising, but individual responses vary and no treatment works for every patient.
How long do the effects of one day TMS last?
In the primary 2024 case series, approximately 50% of patients showed sustained remission at six months post-treatment, while about 25% experienced some relapse or required retreatment. The durability data is still accumulating, and longer follow-up studies are ongoing.
Is one day TMS covered by insurance?
Standard TMS for qualifying diagnoses is covered by most major insurance plans. One day TMS, being off-label, is generally not covered in the same way and would typically be an out-of-pocket expense. Confirm coverage details directly with your insurer and the clinic before scheduling.
Who should not consider one day TMS?
Anyone with contraindications to standard TMS, including certain metallic implants in or near the skull, a history of seizure disorders, or other specific medical conditions flagged during a clinical evaluation, would also be excluded from one day TMS. A thorough pre-treatment assessment is essential regardless of which TMS format is being considered.