TMS for Addiction: Brain-Based Treatment to Support Recovery
Transcranial Magnetic Stimulation (TMS for addiction) offers a breakthrough method for addressing substance use disorders by applying focused magnetic pulses to brain areas responsible for reward processing, impulse control, and craving responses. These magnetic waves, similar in nature to those used in diagnostic MRI equipment, help recalibrate the neural pathways that have been altered by prolonged substance use.
By addressing these critical brain regions, TMS therapy for addiction works to reduce the intensity of cravings and strengthen self-regulation mechanisms, offering individuals a scientifically-backed complement to traditional recovery programs without relying on additional medications.
Why Choose TMS Addiction Treatment?
Transcranial magnetic stimulation for addiction provides unique benefits for individuals committed to overcoming substance dependence. Here are the key factors that make TMS treatment for addiction worth considering:
- Non-invasive: No incisions, anesthesia, or injections are involved in the process.
- FDA-approved: TMS technology has FDA approval and is backed by clinical research demonstrating its effectiveness.
- Minimal side effects: The treatment is generally well-received, with most patients reporting little to no adverse reactions.
- No downtime: You can maintain your work schedule, family obligations, and recovery meetings without interruption.
- Quick sessions: Each appointment takes 20-40 minutes, fitting seamlessly into a busy recovery schedule.
- Long-lasting results: Many individuals experience sustained reduction in cravings and improved impulse control after completing their treatment series.
What to Expect During Your TMS Therapy for Addiction Session
Before Treatment
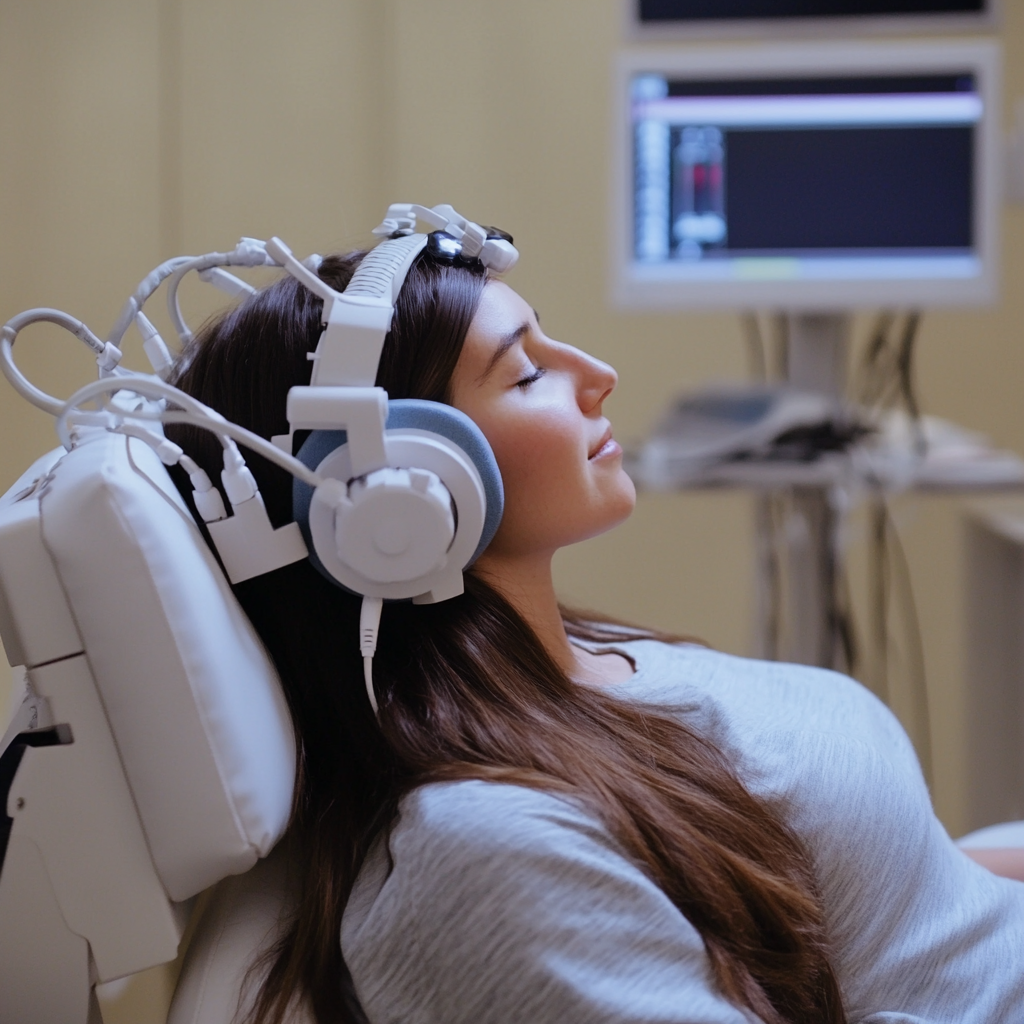
- Upon entering the treatment area, you'll be seated in a comfortable reclining chair where you can relax during the session. A specialized helmet containing the TMS coils will be carefully positioned on your head, targeting the specific brain regions involved in addiction and craving cycles. Your provider may take a few moments to ensure you're comfortable and address any last-minute questions before beginning the session.
During Treatment
- The TMS device delivers targeted magnetic pulses to the neural circuits associated with addiction behaviors. During the process, you'll hear rhythmic clicking sounds produced by the equipment and feel a gentle tapping sensation on your scalp—similar to light fingertip pressure. You might also notice slight, brief muscle contractions in your facial area, which is a normal and harmless occurrence. The magnetic pulses are administered in timed intervals, with each pulse train lasting a few seconds and pauses of 10-20 seconds between sequences to maintain optimal treatment conditions. The entire session typically requires 20 to 40 minutes, depending on your personalized treatment approach and clinical goals.
After Treatment
- Once the session is finished, you're immediately able to resume all normal activities. TMS for addiction doesn't cause drowsiness, confusion, or impaired motor skills, so you can safely drive yourself home or head directly to work, support group meetings, or other commitments.
Frequently Asked Questions About TMS for Addiction
Can TMS for addiction help with alcohol dependence?
Yes. Research shows that transcranial magnetic stimulation for addiction can significantly reduce alcohol cravings by targeting the prefrontal cortex regions involved in decision-making and impulse control. Many patients undergoing TMS therapy for alcohol use disorder report decreased urges to drink and improved ability to resist triggers, making it a valuable addition to comprehensive alcohol recovery programs.
Is TMS for addiction FDA-approved?
Yes. The FDA has cleared TMS therapy for addiction as a safe and effective intervention. The technology has been extensively studied and utilized in clinical settings, with evidence supporting its role in reducing cravings and supporting recovery outcomes.
Does TMS for addiction cause pain or discomfort?
Most individuals undergoing TMS treatment for addiction report minimal discomfort. You may feel a light tapping on your forehead region, similar to gentle drumming. Some patients experience mild scalp sensitivity or brief headaches following treatment, but these symptoms are typically short-lived and manageable.
How soon will I start seeing results from TMS for addiction?
Many people begin noticing a decrease in craving intensity and improved self-control by the 2nd or 3rd week of treatment. For others, the full therapeutic effects may emerge around weeks 4-5. Research demonstrates that transcranial magnetic stimulation for addiction produces meaningful benefits for the majority of patients who complete the recommended treatment course.
Is TMS safe for everyone with addiction?
While TMS addiction treatment has a strong safety record, it’s not suitable for every individual. This therapy is often recommended for those who continue to struggle with cravings despite participating in counseling, recovery programs, or medication-assisted treatment. Individuals with certain conditions, such as epilepsy or metal implants in the head, require careful screening by their healthcare provider before starting treatment.
Can I receive TMS if I'm currently in a rehabilitation program?
Absolutely. TMS addiction treatment integrates well with residential rehab, intensive outpatient programs (IOP), and standard outpatient treatment. Many recovery centers recognize TMS as a complementary therapy that addresses the neurological components of addiction while patients engage in counseling, group therapy, and skills development. Your treatment team will coordinate care to ensure TMS sessions fit within your comprehensive recovery plan.
How much does TMS for addiction cost and is it covered by insurance?
The cost of TMS therapy for addiction depends on your location, treatment duration, and facility. Insurance coverage for TMS is becoming more common, especially for patients who haven’t responded adequately to conventional treatment methods. Our billing specialists will help verify your insurance benefits and explain any out-of-pocket expenses.
Start Your Journey to Relief with TMS for Addiction
If you’re ready to strengthen your recovery and reduce the hold that cravings have on your life, schedule a complimentary consultation with our knowledgeable team today. Let us show you how TMS treatment for addiction can provide the neurological support you need to build a healthier, substance-free future.
Get In Touch With Us
info@lifequalitytms.com
(718) 400-0867
(718) 4000-TMS
26 Court St., Ste 808
Brooklyn, NY 11201