The following article provides a comprehensive look at the regulatory standing of Transcranial Magnetic Stimulation. By explaining the nuances of the question, “Is TMS FDA approved?” and detailing the specific conditions it is authorized to treat, we aim to guide patients and families toward an informed perspective on this evolving psychiatric tool.
The world of mental health treatment often feels like a search for a missing piece of a puzzle. For years, the standard approach has relied heavily on chemistry—specifically, the use of medications to alter brain function. While effective for many, there remains a significant group of people for whom pills simply aren’t enough.
Transcranial Magnetic Stimulation (TMS) has emerged as a landmark alternative, offering a biological, non-invasive way to stimulate the brain without the systemic side effects of pharmaceuticals. Because it represents such a departure from traditional methods, understanding the regulatory status of the technology is a vital part of the conversation.
The Significance of TMS FDA Approval
In the medical community, a label of being FDA-approved carries immense weight. It is the gold standard of legitimacy, indicating that a treatment has undergone rigorous clinical trials to prove it is both safe and effective for its intended use. For patients, knowing that a technology is TMS FDA-approved means they are choosing a therapy that has been vetted by the highest regulatory body in the United States, providing a level of confidence that “alternative” therapies often lack.
What TMS FDA Approval Means for Patients
When a device is TMS FDA-approved, it moves from the realm of experimental research into the world of mainstream medicine. This transition is what typically triggers insurance companies to begin covering the treatment.
Without this official nod, many life-changing therapies remain out of reach for the average person due to high out-of-pocket costs. Furthermore, the approval ensures that the machines being used in clinics are standardized, calibrated, and operated under strict safety protocols.
TMS FDA Approval Timeline and Milestones
The journey of TMS through the regulatory system has been one of steady expansion. It first made headlines in 2008 when the FDA cleared the first TMS device for the treatment of Major Depressive Disorder (MDD) in adults who had not found relief through medication. This was a turning point for psychiatry. Since then, the milestones have continued:
- 2013: Approval expanded to include certain types of deep TMS technology.
- 2018: The FDA cleared TMS as an adjunct treatment for Obsessive-Compulsive Disorder (OCD).
- 2020: Approval was granted for smoking cessation, showing that the technology can help with addiction-related pathways.
- 2021-2022: The focus shifted toward speed and personalization, with the approval of accelerated protocols (like SAINT) that shortened treatment times from weeks to just days.
- 2024-2025: Regulatory clearances expanded to include adolescent patients (ages 15-21), addressing a growing mental health crisis in younger populations.
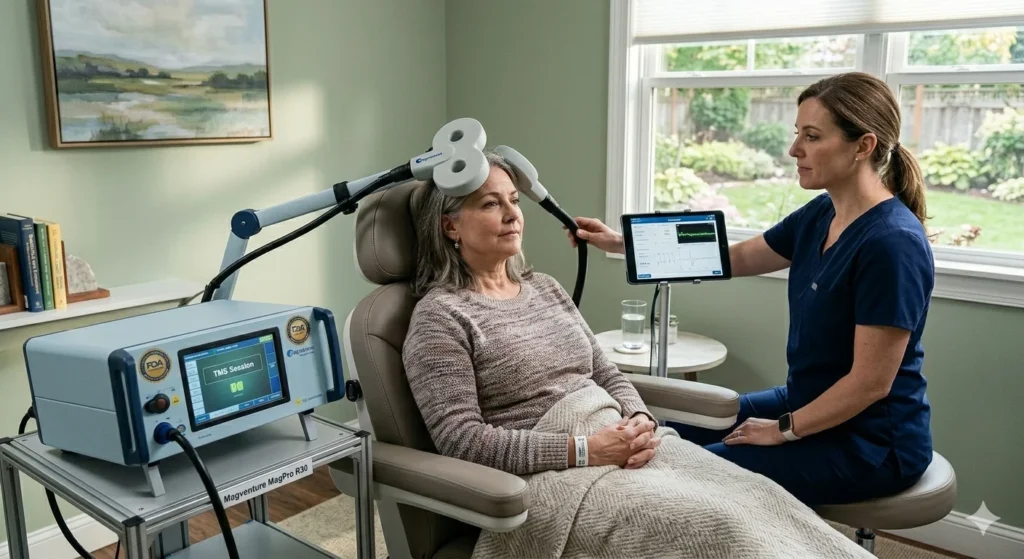
TMS FDA Approved for Depression
The most established use case for this technology is undoubtedly for treatment-resistant depression. For a long time, the only options after medication failure were more medications or highly invasive procedures like Electroconvulsive Therapy (ECT). The fact that the technology is TMS FDA-approved for this specific use case has given millions of people a middle path—one that is effective but doesn’t require anesthesia or cause memory loss.
How TMS Works for Depression
Depression is often associated with underactivity in the prefrontal cortex, the part of the brain that regulates mood. A TMS FDA-approved device uses magnetic pulses to induce a small electrical current in these specific areas. Over a series of sessions, this stimulation helps “wake up” the neurons, improving the brain’s ability to regulate mood naturally. It is less like a drug and more like a workout for the brain’s neural pathways.
Is TMS FDA-approved for Anxiety?
This is one of the most common questions from patients today. Anxiety and depression often walk hand-in-hand, so it is natural to wonder if the same magnetic pulses can calm an overactive mind. The answer is nuanced. While the technology is TMS FDA approved for “anxious depression” (depression with comorbid anxiety symptoms), it does not yet have a standalone approval for Generalized Anxiety Disorder (GAD).
TMS FDA Approval for Anxiety Disorders
In 2021, a major milestone was reached when the FDA cleared certain deep TMS devices for the treatment of anxious depression. This means that if you struggle with depression and also feel high levels of anxiety, the treatment is officially recognized as a valid option for you.
For those seeking treatment purely for GAD or Panic Disorder, many providers offer TMS “off-label,” which is a common and legal medical practice, though insurance coverage for these specific cases can be more difficult to secure.
Can TMS Help with Anxiety?
Clinical research suggests that TMS can indeed be highly effective for anxiety. By targeting different areas of the brain—sometimes the right side of the prefrontal cortex rather than the left—clinicians can help dampen the “fight or flight” response. The reason the question “Is TMS FDA approved for anxiety?” doesn’t have a simple “yes” for standalone GAD is simply that the clinical trials are still ongoing to meet the FDA’s strict data requirements for that specific label.
The Expanded Use of TMS FDA Approval
The story of TMS is not yet finished. As our understanding of brain mapping improves, the list of conditions that are TMS FDA-approved is expected to grow. We are moving toward an era of personalized medicine where a psychiatrist might “prescribe” a specific magnetic frequency based on a patient’s unique brain scan.
TMS for Other Mental Health Conditions
Currently, research is heavily focused on conditions like Post-Traumatic Stress Disorder (PTSD) and Bipolar Disorder. In many studies, TMS has shown a remarkable ability to help veterans and trauma survivors regain a sense of calm. While these uses are not yet fully TMS FDA approved, the preliminary results are promising enough that many specialized clinics are already incorporating these protocols into their work.
Potential Future FDA Approvals for TMS
Looking ahead toward the end of 2026 and beyond, we may see approvals for neurological issues such as early-stage Alzheimer’s or stroke recovery. The core principle—that magnetic fields can safely stimulate or inhibit neural activity—has potential applications across the entire spectrum of brain health. The regulatory process is slow by design to ensure safety, but the momentum is undeniable.
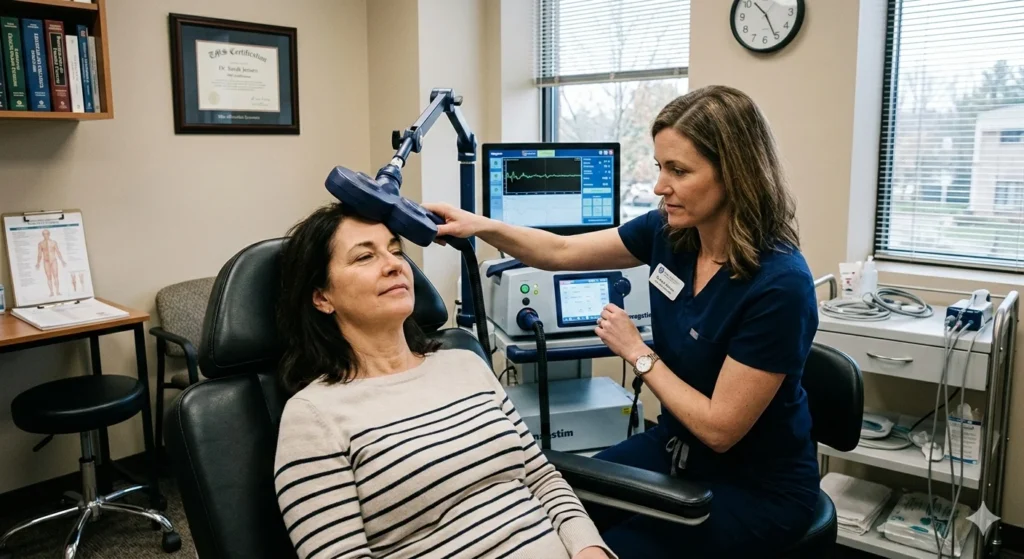
What to Expect During TMS Therapy
If you are considering a treatment that is TMS FDA-approved, it helps to know what the day-to-day reality looks like. This is not a “one and done” procedure, but rather a cumulative process of brain retraining.
The Treatment Process
A typical course involves sessions five days a week for about six weeks. You sit in a comfortable chair while a magnetic coil is placed against your head. You will hear a clicking sound—much like a woodpecker tapping on a helmet—and feel a light tapping sensation on your scalp.
You are awake and alert the whole time; you can talk to the technician, watch a show, or simply listen to music. Because the technology is TMS FDA-approved as a non-systemic treatment, there is no downtime. You can drive yourself home or back to work immediately after the session.
Side Effects and Monitoring
One of the main reasons patients seek out TMS FDA-approved care is the lack of harsh side effects. There is no weight gain, no sexual dysfunction, and no “brain fog” often associated with pills. The most common side effect is a mild, temporary headache or some scalp discomfort where the coil was placed.
These usually fade within the first week of treatment as the body adjusts. Your provider will monitor your progress closely, adjusting the intensity of the magnetic pulses to ensure you are comfortable throughout the process.
Conclusion: A New Era of Legitimacy
The question “Is TMS FDA approved?” is more than just a regulatory inquiry; it is a question about the future of how we treat the human mind. The TMS FDA approval has successfully pulled neuromodulation out of the shadows of “experimental” medicine and placed it firmly in the center of modern psychiatric care. It has provided a much-needed lifeline for those who felt they had run out of options.
As we see more conditions become TMS FDA-approved, the stigma surrounding brain stimulation continues to dissolve. We are learning that the brain, like any other organ, sometimes needs a physical intervention to function at its best.
Whether it is for depression, OCD, or the potential for standalone anxiety treatment in the future, TMS stands as a testament to how far we have come in our ability to heal without a prescription pad. For anyone standing at the crossroads of their mental health journey, the growing role of TMS therapy offers a powerful, proven, and increasingly accessible path toward a brighter horizon.
