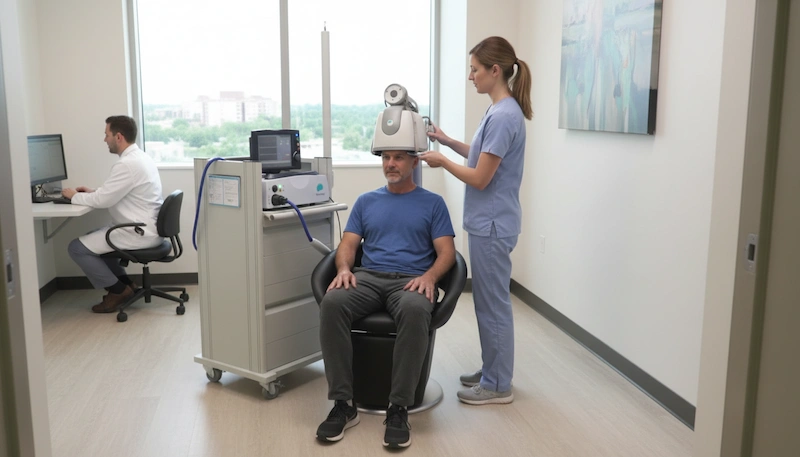
When people start researching TMS therapy, they often assume all devices work the same way. In reality, the technology behind the machine matters enormously – it determines how deep the magnetic field reaches, which brain structures can be targeted, and ultimately which conditions can be treated.
BrainsWay Deep TMS stands apart from conventional TMS in each of these respects. It is the most clinically validated deep brain stimulation platform in the field, and it is the system used at LifeQuality TMS in Brooklyn. Here is what makes it different, and why that difference matters for patients.
What Is BrainsWay Deep TMS?
BrainsWay dTMS is a non-invasive brain stimulation technology that uses a patented H-coil design, housed in a helmet-like device, to deliver magnetic pulses deeper and more broadly into the brain than conventional figure-eight coils. While standard TMS reaches only surface-level cortical tissue, BrainsWay Deep TMS technology reaches subcortical structures – the deeper brain regions that play a central role in mood regulation, compulsive behavior, and addiction.
BrainsWay is the first and only TMS company to obtain three FDA-cleared indications backed by pivotal clinical studies demonstrating clinically proven efficacy. Those indications are major depressive disorder (including anxious depression), obsessive-compulsive disorder, and smoking addiction. No other TMS manufacturer holds this combination of FDA-cleared indications supported by its own pivotal trial data.
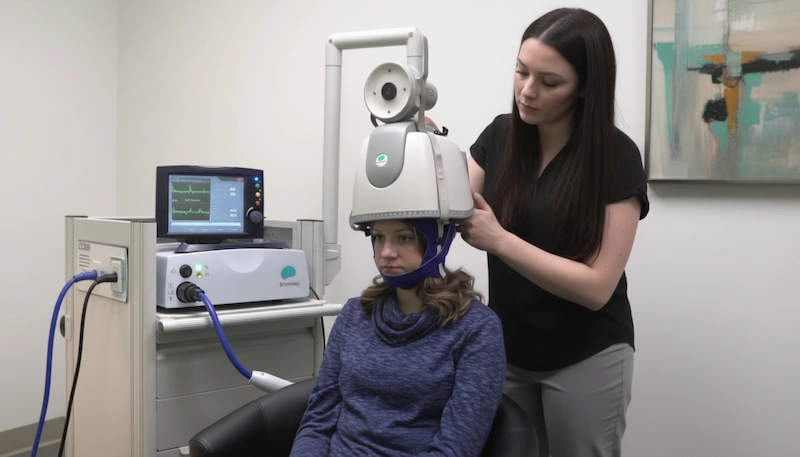
The Science Behind the H-Coil
A Technology Born at the NIH
The magnetic coil technology at the heart of BrainsWay Deep TMS – called the H-coil – was developed in the late 1990s and early 2000s through research at the U.S. National Institutes of Health. The H-coil’s flexible winding configuration generates a magnetic field that penetrates deeper into brain tissue than the figure-eight coils used in standard TMS systems, while also covering a broader stimulation area.
Comparative electric field mapping has shown that H-coils can stimulate up to five times more brain volume than figure-eight coils. This expanded reach is not about delivering more stimulation indiscriminately – it is about accessing brain circuits that are structurally deeper and have previously been difficult to target non-invasively.
Multiple Coils for Multiple Conditions
BrainsWay currently holds 16 patented H-coil configurations, each designed to target specific brain regions associated with different disorders. The two most clinically relevant for patients today are:
- The H1-coil: Targets the bilateral prefrontal cortex with a preference for the left dorsolateral prefrontal cortex (DLPFC). This is the primary coil used for depression treatment and was the first to receive FDA clearance in 2013.
- The H7-coil: Designed to stimulate the medial prefrontal cortex and anterior cingulate cortex – the brain structures most closely linked to OCD. This coil received FDA clearance for OCD in 2018, making BrainsWay Deep TMS the first non-invasive device ever cleared to treat the condition.
FDA-Cleared Indications: What BrainsWay Treats
Depression and Anxious Depression
BrainsWay Deep TMS therapy for depression is backed by robust multicenter clinical data. A large post-marketing study involving over 1,300 patients with treatment-resistant depression found that 82% experienced a meaningful response to treatment, and 65% achieved full remission. These are real-world results from patients who had already failed multiple medications – not a controlled trial with carefully filtered participants.
In a further expansion of the depression indication, BrainsWay received FDA clearance for its Deep TMS H7-coil to treat adults suffering from MDD and depression including those with comorbid anxiety symptoms, commonly known as anxious depression. This addresses a significant clinical gap: many people with depression also experience elevated anxiety, and this dual-indication clearance reflects BrainsWay’s growing evidence base for treating the full spectrum of depressive presentations.
OCD
The OCD indication for BrainsWay Deep TMS therapy is clinically distinctive. Because OCD involves overactivity in circuits that run deeper than what surface-level TMS can reliably reach, the H7-coil’s deeper field is particularly well suited to this condition. A peer-reviewed multicenter clinical study found Deep TMS to be a highly effective OCD treatment, with more than one in three treatment-resistant OCD patients achieving full response, greatly improving their quality of life.
Real-world post-marketing data from over 200 patients across 22 clinical sites found that nearly 60% of patients achieved response after 29 sessions, and over 70% of patients reached first response after an average of 18.5 Deep TMS sessions or 31.6 days.
Smoking Cessation
In 2020, BrainsWay received FDA clearance for its H4-coil for short-term smoking cessation in adults. This makes BrainsWay the only TMS system cleared for an addiction indication. For patients seeking a medication-free approach to quitting smoking, this represents a meaningful additional option within the same treatment platform.

BrainsWay Accelerated TMS and the SWIFT Protocol
A Faster Path to Relief
One of the most significant recent developments in BrainsWay Deep TMS technology is the FDA-cleared SWIFT protocol – an acronym for Short-course with Intrinsic Field Targeting. BrainsWay accelerated TMS via SWIFT condenses the traditional 20-session acute treatment phase into just six half-day visits, followed by weekly maintenance sessions for four weeks.
This is a substantial shift in how BrainsWay Deep TMS therapy is delivered. The standard protocol requires daily clinic visits five days a week for several weeks. The SWIFT Brainsway protocol achieves comparable outcomes in a fraction of the time – without requiring functional MRI or expensive neuronavigation equipment, unlike some other accelerated approaches.
What the Clinical Data Show
In a large, randomized, multisite non-inferiority trial, patients treated with BrainsWay’s accelerated protocol achieved an 87.8% response rate and a 78.0% remission rate, with a median time to remission of 21 days versus 28 days for the standard protocol. Those results held consistently across patient subgroups, including individuals with moderate-to-severe depression and comorbid anxiety.
Patient-reported outcomes reinforce the clinical data. By six weeks, quality-of-life scores improved by approximately 32 percentage points from baseline. The proportion of patients reporting severe functional impairment declined from 85-100% at baseline to 15-17% at the end of treatment, and approximately 60% reached normal-range functioning by the end of treatment.
The SWIFT protocol’s accelerated schedule also addresses one of the most common practical barriers to completing TMS treatment: the burden of months of daily clinic visits. By reducing the acute phase by approximately 70%, it makes BrainsWay deep TMS therapy meaningfully more accessible for working adults, caregivers, and anyone whose schedule makes a multi-week daily commitment difficult.
How BrainsWay Compares to Other TMS Systems
| Feature | Standard rTMS | BrainsWay Deep TMS |
| Coil type | Figure-eight | Patented H-coil (multiple configurations) |
| Brain depth and coverage | Surface cortex only | Deeper + broader field (up to 5x more volume) |
| FDA-cleared indications | Depression | Depression, Anxious Depression, OCD, Smoking Cessation |
| Accelerated protocol | Varies by device | SWIFT – FDA-cleared, no fMRI required |
| Insurance coverage | Widely covered | Widely covered (standard); SWIFT coverage expanding |
| Age range | Adults | Adults and adolescents (15+) for depression |
BrainsWay at LifeQuality TMS: What to Expect
At LifeQuality TMS in Downtown Brooklyn, we use the BrainsWay Deep TMS system to treat depression, anxious depression, and OCD. Every patient begins with a psychiatric evaluation by one of our licensed psychiatrists, who will determine whether BrainsWay Deep TMS is clinically appropriate for you and which protocol best fits your diagnosis, schedule, and treatment history.
The treatment itself is straightforward. Sessions take approximately 20 minutes. You remain fully awake and seated in a comfortable chair throughout, and you can return to your normal daily activities – including work and driving – immediately afterward. There is no sedation, no surgery, and no recovery time required.
We handle all insurance authorizations on your behalf. BrainsWay Deep TMS is covered by most major insurance plans, including Aetna, Blue Cross Blue Shield, Medicare, Cigna, United Healthcare, and many others. Our team works directly with your insurer to obtain the necessary approvals before your first session.
BrainsWay Deep TMS: Conclusion
BrainsWay Deep TMS represents the most clinically validated and broadly cleared non-invasive brain stimulation platform available today. Its H-coil technology reaches deeper and wider than conventional TMS, its three FDA-cleared indications are each backed by pivotal clinical trials, and the new SWIFT accelerated protocol is making treatment faster and more accessible without compromising outcomes.
For patients in Brooklyn and across New York City who are looking for a proven, medication-free path to relief from depression, OCD, or the need to quit smoking, BrainsWay Deep TMS at LifeQuality TMS offers all of this – covered by most major insurers. Schedule a consultation to take the first step.
Frequently Asked Questions
1. What makes BrainsWay Deep TMS different from standard TMS?
The key difference is the H-coil, which generates a deeper and broader magnetic field than the figure-eight coils used in conventional TMS systems. This allows BrainsWay dTMS to reach subcortical brain regions that standard TMS cannot target effectively, which is why it holds FDA clearance for OCD and smoking cessation in addition to depression – indications that require stimulating deeper neural circuits.
2. Is BrainsWay Deep TMS therapy covered by insurance?
Yes, for its FDA-cleared indications. BrainsWay Deep TMS is covered by most major insurance plans in the United States. LifeQuality TMS works with Aetna, Medicare, Blue Cross Blue Shield, Cigna, United Healthcare, and many others.
3. What is the SWIFT BrainsWay protocol, and how does it differ from the standard protocol?
SWIFT (Short-course with Intrinsic Field Targeting) is BrainsWay’s FDA-cleared accelerated deep TMS protocol. Rather than five daily sessions per week for several weeks, SWIFT delivers five 10-minute sessions per day over six days, followed by weekly maintenance visits for four weeks.
4. How many sessions does BrainsWay Deep TMS typically require?
The standard protocol involves daily sessions five days per week for approximately four to six weeks, totaling 20-29 sessions in the acute phase. The SWIFT accelerated protocol reduces this to six half-day visits during the acute phase. Your provider will recommend the appropriate protocol based on your diagnosis and clinical history.
5. Is BrainsWay Deep TMS safe? Are there side effects?
BrainsWay Deep TMS has a well-established safety record across more than a decade of clinical use. It is non-invasive and requires no anesthesia, surgery, or sedation. The most commonly reported side effects are mild scalp discomfort or headache during or shortly after sessions, which typically decrease as treatment progresses.